 |



  

  

  




PB272 (neratinib) is a potent irreversible tyrosine kinase inhibitor, or TKI, that blocks signal transduction through the epidermal growth factor receptors, or EGFRs, HER1, HER2 and HER4. We believe neratinib has clinical application in the treatment of several cancers, including breast cancer and gastric cancer. Our initial focus is on the development of neratinib as an oral treatment for patients with HER2-positive breast cancer.
Data were published in the Journal of Clinical Oncology in March 2010 from a Phase II trial of neratinib administered as a single agent to patients with HER2+ metastatic breast cancer. The trial involved a total of 136 patients, 66 of whom had received prior treatment with approved anticancer drug Herceptin (trastuzumab), and 70 of whom had not received prior treatment with Herceptin (trastuzumab). The results of the study showed that neratinib was reasonably well tolerated among both the pretreated patients and the patients who had not received prior treatment with trastuzumab.
The efficacy results from the trial showed that the objective response rate was 24% for patients who had received prior trastuzumab treatment and was 56% for patients with no prior trastuzumab treatment. Furthermore, the median progression free survival was shown to be 22.3 weeks for the patients who had received prior trastuzumab and 39.6 weeks for the patients who had not received prior trastuzumab, as show on the table below.
Neratinib is currently being tested in a number of clinical trials as a neoadjuvant therapy for patients with HER2+ breast cancer and as a treatment for patients with metastatic HER2+ breast cancer. A list of clinical trials for neratinib can be found at:
http://www.clinicaltrials.gov/ct2/results?term=Puma+Biotechnology&pg=1
Neratinib Phase II MBC
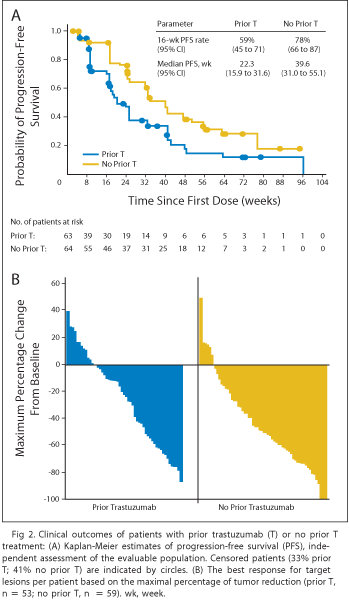
Source: Journal of Clinical Oncology, March 10, 2010.


|


Puma Biotechnology, Inc.
10880 Wilshire Blvd., Suite 2150
Los Angeles, CA 90024
424-248-6500 Main
424-248-6501 Fax

|




